Can atoms and molecules move?PROBLEM:
PREDICTION:
PROCEDURE:
MATERIALS: beaker, 2 electrodes (one copper), items to plate, wire, battery, copper sulfate (CuSO4) solution
EXERCISE I. Set up the equipment as noted in the diagram. When dissolved in water, the copper sulfate (CuSO4) breaks down into copper ions (Cu2+) and sulfate ions (SO42-). When an electric current is applied to the solution, the Cu2+ ions in solution will travel to the negative (-) electrode. Label the diagram below.
+ hook up copper - hook up zinc or whatever you want to plate
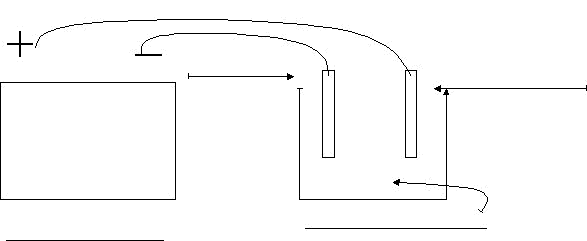
What is the chemical formula for copper sulfate?
What does the copper sulfate break into when it is in the water?
EXERCISE II. Experiment with electroplating other items and record your observations.
|
item |
observation - what happens? |
How do the copper ions move?CONCLUSION: