BACKGROUND:
An electric current is the flow of
electric charge which transports energy from one place to another. It is
measured in amperes, where 1 ampere is the flow of 6 1/4 billion-billion
electrons (or protons) per second. However, no current exists unless
there is a complete pathway or circuit through which electrons may flow.
The flow of electricity through a circuit can be stopped by breaking or
opening the circuit with a switch. By closing the circuit the flow of
electricity can be restored.
There are two possible types of electric flow, direct and alternating current. Direct current means that the flow of charges is in one direction. A battery produces direct current (DC) because there is no way to change the + and - you
see on the battery. Alternating current (AC) has electrons in the circuit that quickly move first in one direction and then in the opposite direction, alternating back and forth between relatively fixed positions. When you use a transformer, you are using AC. Many calculators, cellular phones and other common items use an AC adapter or transformer which helps extend the longevity of the item. The worksheet shows direct current.
Benjamin Franklin was the first to notice the direction of
electrical flow (from the smooth wax to the rough wool), he set a precedent for electrical notation that exists to this
day. However, today we know electrons are displaced from the wool to the wax, not from the wax to the wool. This is why electrons are said to have a negative
charge because Franklin thought electric charge moved in the opposite direction that it actually does, and so objects he called "negative"
have a surplus of electrons.
By the time the true direction of electron flow was discovered, the nomenclature of "positive" and "negative" had already been so well established in the scientific community that no effort was made to change
it. However, many engineers decided to retain the old concept of electricity with "positive" referring to a surplus of charge, and label charge flow (current) accordingly. This
is known as conventional flow notation.
Electric currents travel easily
through some materials like metal and poorly through others like
plastic. The substances that electricity can easily flow through are
called conductors, those that do not allow electricity to flow are
called non-conductors. Some substances restrict the flow of electricity
and these are called semiconductors. Silicon is the most famous
semiconductor used in the computer industry.
PROCEDURE:
-
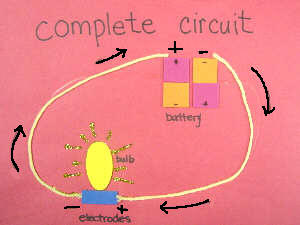 When a circuit is
complete, as shown on the student's worksheet, the current flows from a
region with excess or high number of electrons (the negative pole of a
battery) to a region with relatively few or low electrons (the positive
pole of a battery).
When a circuit is
complete, as shown on the student's worksheet, the current flows from a
region with excess or high number of electrons (the negative pole of a
battery) to a region with relatively few or low electrons (the positive
pole of a battery).
-
Use art to illustrate the flow of electrons in a
closed circuit. In the picture it shows how paint, glitter,
and foam cut outs can reinforce the movement of
electrons.
-
Batteries are the
simplest source of power for DC. Batteries are chemical apparatuses that
allow "electrons" to be generated for use later on. Each
battery has either 1 cell or multiples of cells. Each cell is 1.5 volts.
Many of the common batteries are composed only of 1 cell. Whether the
battery is an A, AA, B, C, or D, size determines how long the battery
will last. It does not refer to the voltage. A six-volt battery has 4
unit cells stacked in a series. You may want to show students different
batteries and a transformers.
-
ANSWERS:
- Current flows from the
negative to the positive.
- A switch is closed when it completes the
circuit and allows electrons to flow continuous.
- An electric circuit
is when the circuit is closed and the electrons are flowing. Display a
battery or other source that can generate electrons.