BACKGROUND:
Why are the oceans so salty?
Rain on land causes rivers to wash salt minerals from the land and carry
them to the oceans. Rivers not only carry salts to the ocean but
also many other elements. In fact, the oceans contain gold and silver!
If you calculate how much salt there is in the ocean, there would be enough
salt to give millions of boxes of salt to each person living in the United
States. That's a lot of salt! Salt is a very important commodity
to people around the world. Before refrigerators, people had to salt
their food in order to preserve it. Salt is important for preserving
food and organisms need a balance of salt in their bodies in order to stay
healthy.
PROCEDURE:
- Compare the periodic table placemats with
the elements that are in seawater figure. Students can
then discover the components of salt water. Go over what each of the major
constituent symbols including:
Na = sodium
Mg = magnesium
K = potassium
Ca = calcium
H = hydrogen
C = carbon
O = oxygen
S = sulfur
Cl = chlorine
Br
= bromine. |
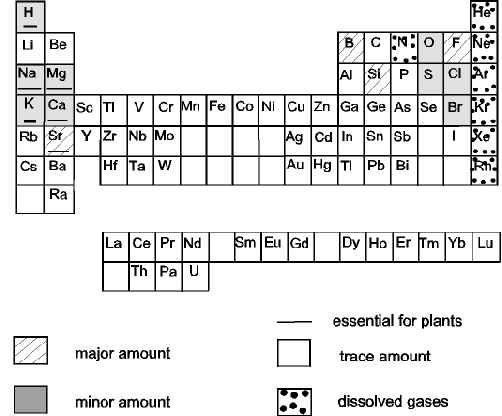
Elements in seawater.
- Use the worksheet to illustrate that salt
water is made during the movement of water on land. The diagram shows areas
where salt is added to the oceans by volcanoes, water erosion of the land, and
evaporation of pure water which leaves the salts behind.
- ANSWERS:
- Clouds do not have salt in
them. Only water can evaporate.
- Yes, large seas and old lakes have salt in
them. Mediterranean Sea and Salt Lake are just a few. Students need to look at
the globe.
- Sources of salt: volcano and land; evaporation is occurring over
the water; erosion is occurring on land and in parts of the ocean that moves.
|