|
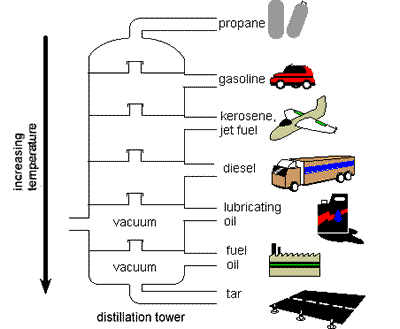 As new sources of crude oil became available, scientists started to
look for other products. Crude oil needs to be refined to produce
products, unlike natural gas which could be used untreated. The heating
of crude causes different products to be distilled in a tower. Crude oil
which is just hydrocarbons (hydrogen and carbon) have different boiling
points. Kerosene and diesel require higher boiling points than gasoline,
which requires lower temperatures. As new sources of crude oil became available, scientists started to
look for other products. Crude oil needs to be refined to produce
products, unlike natural gas which could be used untreated. The heating
of crude causes different products to be distilled in a tower. Crude oil
which is just hydrocarbons (hydrogen and carbon) have different boiling
points. Kerosene and diesel require higher boiling points than gasoline,
which requires lower temperatures.
This has to do with the ability of hydrocarbons to form chains
of hydrogens and carbons. Different lengths of the chains will create
different products through distillation. It was also realized that
distillation in a vacuum will also cause chains to produce new products.
Chemical processes like "cracking" of crude oil breaks long
hydrocarbons into shorter chains which produce gasoline.
"Reforming" of different by-products with heat, pressure and
catalysts, will make chains start chains to create branches. These will
also produce new products.
The products from petroleum seemed endless as scientists started to
understand the chemistry of the hydrocarbons and how they react under
different chemical techniques.
|